Healthspan Weekly — Mar 09, 2026
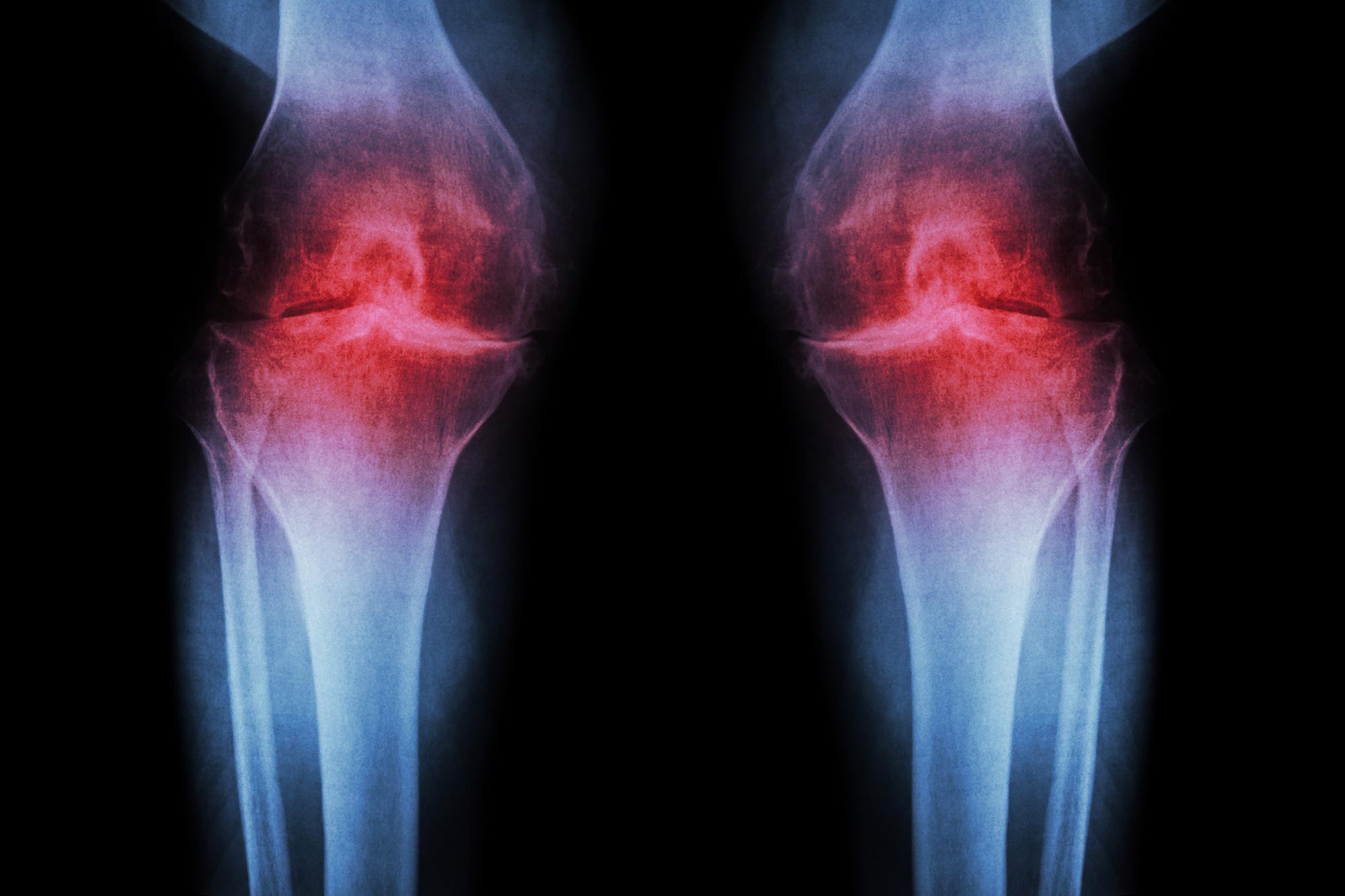
Photo: scitechdaily.com
Week of March 9, 2026
The Big Picture
The machinery of aging is becoming legible in ways it wasn't six months ago. This week, independent teams zeroed in on specific molecular mechanisms — a protein quietly damaging your knees, a Parkinson's molecule accelerating Alzheimer's almost exclusively in women, ribosomes that get worse at their jobs with every passing year — and in several cases, they showed you could actually intervene. Meanwhile, the tools to measure aging got meaningfully sharper, the first human trial of epigenetic reprogramming officially cleared the FDA, and $144 million in federal funding landed to accelerate the whole pipeline. The theme: we're crossing from "aging is complicated" to "aging has addresses, and we're starting to knock on doors."
This Week's Stories
The Protein That's Been Quietly Damaging Your Knees — And How to Stop It
If you've ever been told your cartilage is just "worn out" — nothing to do but manage the pain or eventually get a joint replaced — Stanford gave you a reason to push back on that story this week.
Published in Science, the study found that blocking a protein called 15-PGDH can regenerate cartilage that naturally wears away in the knees of aging mice. The researchers call 15-PGDH a "gerozyme" — a master regulator of aging whose levels roughly double in knee cartilage over a lifetime. Think of it as a volume dial for tissue decline that gets slowly cranked up as you age.
When they blocked it with a small-molecule drug, cartilage that had become thin and dysfunctional thickened across the joint surface. Crucially, the regenerated tissue was hyaline cartilage — the real, smooth, load-bearing stuff — not the scar-like fibrocartilage the body typically produces in repair attempts. In injury models mimicking ACL tears (which lead to osteoarthritis in about half of patients within 15 years), a four-week course of injections dramatically reduced arthritis development.
The human signal is what elevates this beyond a typical mouse study. Samples of human tissue from knee replacement surgeries — both the extracellular scaffolding and the cartilage-generating chondrocyte cells — also responded to the treatment by making new, functional cartilage. And an oral version of the compound is already in clinical trials for age-related muscle weakness, meaning the first human safety hurdles have been cleared.
Independently, Northwestern reported an injectable bioactive compound that stimulated joint repair and reduced inflammation in mice through a different mechanism. Two teams, two approaches, both credible — which adds confidence that regenerative approaches to osteoarthritis are now a real clinical direction.
Osteoarthritis affects one in five American adults and costs $65 billion a year. Until now, no drug could slow, stop, or reverse it. Watch for whether the ongoing muscle-weakness trial expands its endpoints to include joint tissue.
A Parkinson's Protein Linked to 20-Fold Faster Alzheimer's Tau Accumulation in Women

Source: insideprecisionmedicine.com
Here's a finding that should change how we think about brain aging in women — and how we design the trials meant to protect it.
Researchers studying 415 participants in the Alzheimer's Disease Neuroimaging Initiative examined alpha-synuclein — the protein that misfolds in Parkinson's disease — which can appear at abnormal levels in Alzheimer's patients even without a Parkinson's diagnosis. About 17% of participants tested positive for misfolded alpha-synuclein aggregates in cerebrospinal fluid at baseline.
Then they tracked tau accumulation over time. Tau is the protein that tangles and drives cognitive decline in Alzheimer's. Among men, alpha-synuclein didn't change much. Among women, it was associated with more than 20-fold higher tau accumulation rates during the study's follow-up period compared to men with the same pathology.
Twenty-fold. That's not a rounding error — that's a different disease in a different body.
"This opens an entirely new direction for understanding why women bear a disproportionate burden of dementia," said lead author Elijah Mak of Mayo Clinic. Women already experience Alzheimer's at higher rates and see faster cognitive decline — this finding may explain a significant chunk of why.
This raises concern that clinical trials treating men and women as equivalent in their Alzheimer's pathology may miss the most vulnerable patients. Women with any family history of dementia should be asking their doctors whether alpha-synuclein testing belongs in their picture.
The First Human Trial to Reverse Epigenetic Aging Has Received FDA Clearance

Source: longevity.technology
For the first time, partial epigenetic reprogramming — the lab technique that can reset some aspects of cellular age — has received FDA clearance to move into people.
Life Biosciences received FDA clearance to start a Phase 1 trial of ER-100, a gene-therapy approach delivering a trio of Yamanaka factors (Oct4, Sox2, Klf4 — the transcription factors that can reprogram adult cells toward a younger state) to test safety and potential vision-restoring effects in people with optic neuropathies. The eye was chosen for good reasons: local delivery reduces systemic risk, and vision outcomes are measurable.
But the broader implication is what matters. This protocol will directly test whether a factory-reset-like intervention can be performed safely in humans without erasing cell identity — the central fear that has kept reprogramming in animal labs. If partial reprogramming can be controlled in people, it opens a clinical pathway for testing rejuvenation across tissues. The field will be watching enrollment, safety readouts, and any off-target gene-expression changes with extraordinary attention.
This is a watershed. Not because it will cure anything tomorrow, but because the question shifts from "can we do this in mice?" to "can we do this in humans safely?"
Exercise Fights Alzheimer's Through Muscle — And We Now Know One Molecular Address
We've known for years that exercise protects the aging brain. What we haven't known is how — specifically enough to do anything with it beyond saying "go for a walk." A study in Nature Aging this week offers one of the cleanest mechanistic answers yet.
Researchers identified that swimming exercise attenuates cognitive decline in Alzheimer's mice by inducing skeletal muscle to release tiny vesicles (think molecular mail) containing a molecule called miR-378a-3p. These vesicles travel to the brain, get absorbed by microglia — the brain's immune cells — and help those cells clean up amyloid plaques.
A complementary study from UCSF pointed to a second route through the liver: exercise increases circulating GPLD1, which tightens the blood-brain barrier, lowers neuroinflammation, and improves cognitive performance in aged mice. Two organs, two mechanisms, one conclusion: exercise biology is a multi-organ conversation with the brain, and each pathway offers distinct drug-target possibilities.
This is still mouse data. But it gives researchers a measurable proxy for whether an exercise intervention is actually working at the brain-aging level — not just on behavioral outcomes. And if you can identify the specific signal, you might eventually deliver it without the exercise. For now, the original treatment remains free and proven.
The old advice — "exercise is good for your brain" — just got a molecular address. Two of them, actually.
A New Biological Age Clock Just Raised the Bar — And Clinics Are Already Shifting
Biological age clocks — tools that estimate how fast your body is aging rather than just counting birthdays — have proliferated rapidly. Some are useful; many are overhyped. A study published this week in Nature Aging may represent a genuine step forward.
Using about 31,000 electronic medical records, researchers developed OMICmAge — a clock that integrates epigenetic markers (chemical modifications to DNA that act like dimmer switches on genes), proteomic data (the proteins your body is currently producing), and metabolomic data (small metabolites flowing through your system) into a single scalable measure. Most clocks are trained on hundreds or a few thousand samples. This one draws on real-world health records at scale, and a separate comparison of 14 aging biomarkers published March 6 found that "pace of aging" measures — which estimate the speed of biological change rather than a snapshot — outperform many classical clocks at predicting mortality and disease.
The practical shift is already happening. Clinics are increasingly adopting pace-based measures like DunedinPACE to track interventions in real time, treating "how fast you're aging" as a clinical vital sign rather than a marketing flourish. A preliminary study this week connected the dots further: individuals who reduced the gap between their biological and chronological age had roughly 23% lower stroke risk during follow-up.
The gap between "research clock" and "clinically validated tool" is closing faster than most people realize.
⚡ What Most People Missed
Your ribosomes age too — and it might be upstream of almost everything. A bioRxiv preprint posted March 8 found that the cell's protein-manufacturing machinery accumulates chemical damage with age, causing ribosomes to stall and pile up like cars on a highway. A separate preprint found ribosome dysfunction appears to precede other aging hallmarks. Multiple labs are now pointing at translation failure as a root cause, not just a symptom — the convergence pattern that historically signals something real.
Fat tissue remembers being obese — even after you lose the weight. A Nature paper resurfacing on Hacker News this week showed adipose tissue retains epigenetic "scars" from past obesity that don't fully reset with weight loss. Directly relevant to the millions on GLP-1 drugs: metabolic history may leave a molecular fingerprint that affects long-term biology even after the number on the scale normalizes.
$144 million just landed to build the infrastructure for aging drugs. ARPA-H awarded funding across seven teams to validate surrogate biomarkers (DNA methylation, wearables, physical function) so regulators can accept shorter trials of geroprotectors like rapamycin and metformin. One funded project: a $22M trial testing an HIV antiretroviral in healthy 60–65-year-olds to quiet age-related inflammation driven by retrotransposons. Money plus measurement plus mechanism — the classic pattern that precedes rapid progress.
A daily multivitamin produced a small but real slowing of epigenetic clocks. A Nature Medicine analysis of the COSMOS trial found two years of daily multivitamin use shifted multiple epigenetic clocks by roughly a few months during the two-year intervention — larger in people who were biologically older at baseline. First large randomized evidence that something this simple can move molecular aging markers at population scale.
"Difficult people" in your life may be literally aging you faster. A PNAS study covered in the Washington Post linked toxic social relationships to accelerated epigenetic aging. Meanwhile, Yale research found positive beliefs about aging were strongly associated with physical and cognitive improvement in older adults. The narrative you live inside — and the people you live inside it with — shows up in your cells.
📅 What to Watch
- If the ongoing 15-PGDH muscle trial expands endpoints to include joint tissue, it signals human cartilage data could arrive years earlier than a standalone trial would allow — the fastest path to a drug for osteoarthritis.
- If Life Biosciences announces enrollment of the first patient in the ER-100 epigenetic reprogramming trial, it becomes the first controlled test of cellular rejuvenation in a living human — expect platform-defining safety readouts to recalibrate investor funding and trial designs.
- If the ribosome-aging preprints replicate in mammalian models, "ribosome rejuvenation" joins mitochondria and epigenetics as a named therapeutic target — and protein quality control moves from niche to central in aging biology.
- If upcoming Alzheimer's trials adopt multi-marker stratification (alpha-synuclein + p-tau217 + sex), treatment effects that have been muddied by mixed cohorts could suddenly become dramatically clearer — especially for women.
- If Buck Institute releases early Healthspan Horizons participant benchmarks this year, we'll see whether combining wearables with molecular data can identify population-scale signals for what actually preserves independence — the kind of evidence that changes insurer and employer behavior.
That's the week. The most underappreciated longevity intervention still isn't a drug — it might be staying meaningfully engaged with people and work that matter to you. But the drugs are getting more interesting by the month.
Nothing here is medical advice. Talk to your doctor before changing anything. We just read everything so you don't have to.